At the forefront of the Pharmaceutical and Life Science Industry
We know your world
Increasing demand for highly specific and targeted drug delivery, associated regulatory requirements and scientific and technological advances are all converging to create ever more challenging conditions for the pharmaceutical and life science markets. Responding to these market drivers we offer a highly competitive design, build and maintain solution that helps realise our client's strategies, bringing products to market first.

Your Objectives - Our Promises
Contamination control strategies and cleanroom processing are critical to quality, security and reproducibility in modern manufacturing processes and production capacity planning. We can help you:
- Ensure commercial viability by helping to build proof of concepts for scale-up and demonstrator facilities
- Guarantee regulatory and pharmaceutical cGLP/cGMP compliance
- Ensure optimum levels of production capacity planning, contamination control and product quality
- Improve product performance and accelerate time to market
- Achieve the total lowest lifecycle of assets and facilities
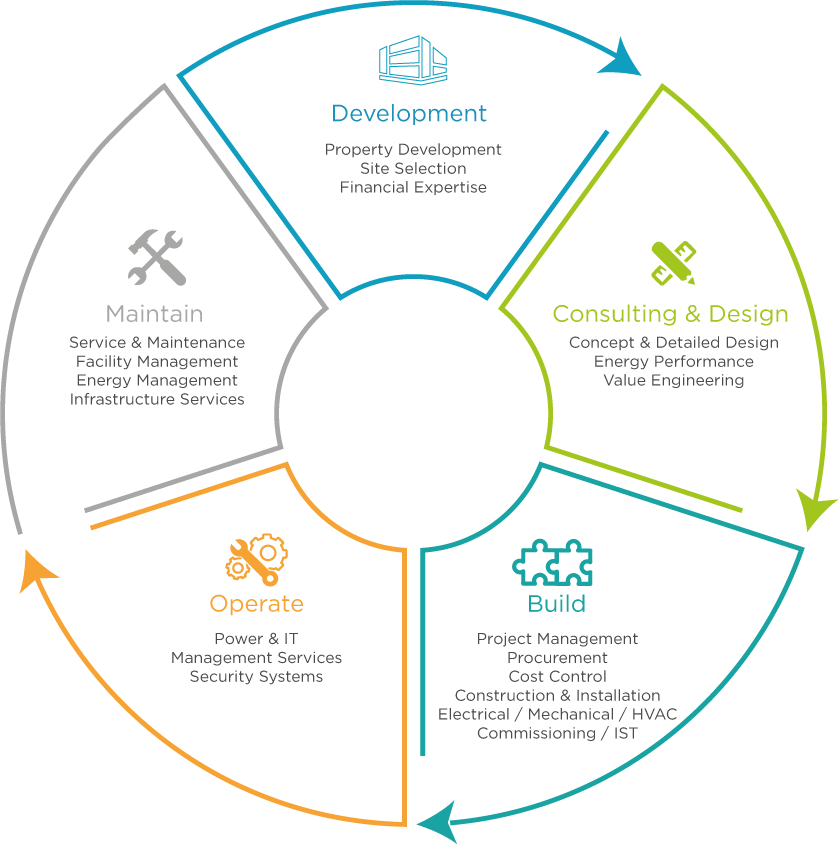
Our Solutions
Understanding our client's product and processes and integrating their vision into the project execution plan from the outset is key to our success in this industry.
Our projects solutions are generated from the “inside-out”, with manufacturing process as the primary consideration, seamlessly integrated and optimised within the facility spatial design, whether retrofit, expansion or new build. This allows us not only to conceptualise and capture your requirements in an accelerated manner, but more importantly, enables the integration of specialist equipment, de-risking interface project construction.
A single point of design, delivery and responsibility with the project team is a tangible benefit our clients appreciate. This approach allows seamless transition from design, construction and validation through to handover with no surprise.
In close collaboration with your team, we can optimise your manufacturing processes for a new or existing product. By improving quality and safety standards, optimising capacity planning, boosting agile continuous manufacturing processes and increasing energy efficiency, we help you to maximise your competitive edge.
We provide a comprehensive solution for a wide range of processes and products across a variety of dosage forms and drug delivery systems including laboratory, scale-up and full-scale manufacturing facilities and associated process plants:
- Warehouses and Cold Storage
- Primary/Bulk API facilities
- Secondary manufacturing and packaging facilities
- Terminal Sterilisation and Aseptic Manufacturing facilities
- High containment facilities
- Biologics facilities
- Cellular Therapy facilities
- Medical device
At the Cutting Edge of Control and Facilities
Your products and needs determine our solution. From early stage consultancy through to contracting capability, we offer a seamless integrated lifecycle solution to support and develop your start-up or growth strategies, driving a wide range of innovative products and processes into the market first with minimal risk and on budget.
From initial concepts through to project design, construction and commissioning, our in-house validation solution covers every stage of your project development. This unique approach ensures that we consistently deliver on our promises with the highest levels of production capacity planning, contamination control, product quality and regulatory compliance. We ensure the continuous incorporation of cutting edge practices and innovation complying with the most stringent requirements and guidelines.
Connect with our team:
Our Commitments - Pharmaceuticals & Lifesciences
Our Commitments

De-risk Project Development
Accompanying you through the whole project development from concept stage to handover
Technical Expertise
Leveraging our engineering design and construction technique to accelerate and realise the vision of our clients
Compliance Strategies Within Regulated Sectors
Ensuring MHRA and FDA regulations are met as required
Lean Operation
Ensuring greater long-term operational efficiency and productivity
Latest news
Find all the topics that make Bouygues Energies & Services news: projects, innovations, public and client events, partnerships, press releases ...
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- Learning at Work Week - Fostering a Culture of Continuous Learning & Development within the Business
-
-
-
-
-
-
-
-
-
-
-
- Bouygues E&S Support for Parkinson's
-
-
-
-
-
- International Women's Day
- Climate Fresks at Bouygues E&S
-
-
-
-
- Investing in our people to train the next generation of talent and further the skills of our employees
-
-
-
-
-
-
-
-
- International Men's Day
-
- Bouygues E&S continues to foster and develop relationships with extremely worthwhile causes.
-
-
-
- Bouygues E&S is taking action to transition our fleet to electric vehicles and installing the infrastructure to support this.
-
-
- Bouygues E&S' fundraises for Wear It Pink.
-
-
-
- Last month, Bouygues E&S hosted our first Apprenticeship Induction Day.
- Bouygues Energies & Services advocates its support for mental health services across the UK and within our business.
-
- Graduates raise over £4,000 for this important cause.
-
-
- This National Inclusion Week, Bouygues E&S are particularly proud of its overarching D&I Champions Network.
-
-
- This European Mobility Week, Bouygues Energies and Services is shining the light on key business initiatives.
- Bouygues E&S recently held its first internal ‘Apprentice of the Year Awards’...
-
-
-
- Bouygues E&S celebrates first-ever TopSite award!
- We are excited to announce the launch of Orbiter™ by Bouygues Energies & Services.
-
-
-
-
- Supporting Digital Transformation for Improved Decision-Making and Building Performance Optimisation
- At BYES, we are committed to championing gender equality.
- By supporting them to achieved their Uptime Institute accreditation
-
-
- The School is the result of a collaboration between clients, contractors and suppliers
-
-
-
-
- International Women’s Day #BreakTheBias
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- Today is ‘Earth Overshoot Day’. This is the point in each year when humans have used up a full year’s worth of resources
- At Bouygues Energies & Services, as well as increasing the electric vehicles in our fleet and using telemetry to enable more efficient driving, we also equip our staff with tips to help reduce their carbon footprint while on the road. If you’re keen to reduce your carbon footprint while driving, here are some key tips that will also save you money on your fuel bill in the process!
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- TO STRENGTHEN ITS EXPERTISE IN INDUSTRY 4.0
-
-
-
-
-
-
-
-
-
-
- Bouygues Energies & Services have helped plant over 235 trees in Malawi with the help of Fruitful Office
-
-
-
-
-
- Earlier this week we spoke to Jo Adams, one of our lead Interior Designers
-
-
-
-
-
-
- Bouygues Energies & Services support Bouygues Construction is continuing to sow its seeds in Australia.
- Each year we hold our internal Sustainability Awards open to all our people within Bouygues & Energies Services UK.
-
- Food sustainability is about culture, health, equity and respect for the planet we live on.
- We see you as more than a number…
- Interview with Adeel Aslam, Technical Director, Bouygues Energies & Services, Contracting
- where we provided a solution to help improve the internal air quality
- 7100 m² of data storage in the heart of Marseille’s port district
-
-
- One for the diary, 11th November 2020, 2pm - 3pm (GMT)
- Provide renewable energy produced from the North Sea Wind Farms to homes across Germany by 2024.
-
-
- Pre-fabrication modular block: The paradigm shift to build a data centre differently?
- A virtual celebration to thank our supply chain
-
-
- Find out why we're prioritising electric vehicles
-
-
- There has always been a huge importance on reducing our Global footprint, to ensure a sustainable future
- A day for employers across the country to advocate their support for responsible employment.
- We have signed a first of its kind deal with Airlite, an innovative paint company
- Top tips to reduce you carbon footprint while working from home
-
- Along with 17 other companies the Data Centre team undertook a number of high-level fundraising activities in aid of CRASH charity.
- On this project we will be using our new innovative modular solution to help achieve the quality, speed to market..
- Sustainability can be a uniting theme and in this context great partnerships can be built between universities and the private sector.
- Leon wins the Sustainability Awards!
- We've helped Manchester Cares get closer to their £10,000 target
-
- We're providing a safe long term solution to protect our healthcare workers
- We’ve contributed to over 23,000 fruit portions, 600 fruit baskets being delivered to NHS hospitals.
- Some of our team members share their experiences during Covid-19.
- We've been recognised for our approach to health and safety
- Well done to our four team members that have made the finals!
- The Most Effective Development to a Wellbeing Strategy
-
- ExCel London on the 11th – 12th March 2020
- Bouygues Energies & Services are Exhibiting at this year’s Data Centre World
-
-
-
-
-
- Project Manager Aaron Yule will lead the team in charge of transforming Ashford's lighting.
-
-
-
-
-
- Gold Award for our continued focus on sustainability
- We're exhibiting at this year’s 2019 ISPE Europe Pharma 4.0 Conference, 20 - 21 November 2019, Manchester, Old Trafford.
- We have successfully renewed our Cyber Essentials Plus certification for the whole organisation.
- Virology Research Laboratory extension for AstraZeneca
-
-
- Bouygues Energies & Services are Exhibiting at this year’s DCD Event, 5 -6th November 2019, Old Billingsgate, London.
- David Carr presents his thought leadership piece.
- VIRTUS is the fastest growing Data Centre provider in the UK.
- We're encouraging our staff to share their work stories on social media.
- Bouygues Energies & Services are pleased to announce they are supporting DATACENTRE.ME!
- We're developing an energy project that will transform St Ives Park & Ride into a renewable energy generator.
- We will be presented the award by HRH Princess Anne
-
- Bouygues Energies & Services get moving with the Virgin Pulse
- Bouygues Energies & Services Data Centre team took part in The 21st Dragon Boat Challenge.
-
-
- We're looking for innovative startups to help create the campus of tomorrow!
- We hosted a construction skills academy for local people in Tower Hamlets.
- We're supporting the special Olympics and the great work it does to support 10,000 athletes.
-
- Brent are saving money, saving carbon and creating a safer place to live and work.
-
- Working with young people means that for us, wellbeing is as much of a priority as security.
- Employees have been busy spreading the festive cheer, with Christmas fundraising.
- We have won the award in recognition of our sustainability initiatives.
- We are collaborating with the whole Bouygues Construction Group in the UK to host Health & Safety Day 2018.
- Bouygues Energies & Services are delighted to announce the successful handover of the VIRTUS LONDON5 facility.
- With Sun'R we have combined our skills to support Lamagistère in its energy transition.
- We continue to grow our successful partnership, with further orders from VIRTUS.
- Axione becomes the 86th member of the altnet cooperative.
- Commercial partnership signed between us and Bosch France
- James Williams tells us about his role at Bouygues Energies & Services as a Process Engineer.
- We are delighted to announce a new contract with VIRTUS Data Centres.
- Sustainability initiative for Chelsea and Westminster NHS Foundation Trust.
- What makes a 'good' data centre?
- Bouygues Energies & Services commits to eliminating all single-use plastics from supply chain.
- What do our apprentices really think?
- with Geology graduate, Connor Cassley.
- Bouygues Energies & Service feature in Innovators magazine's biotech special.
- with Interior Architecture and Design undergraduate, Hollie.
- Graduate Trainee, Chris Nicolaou, talks about his experience working in Procurement.
- Bouygues Energies & Services awarded FM Contract with Slough Borough Council
- Bouygues Energies & Services is delighted to achieve certification of the revised ISO 9001 and ISO 14001.
- Bouygues Energies & Services awarded new contract to continue providing Facilities Management to UAL.
- Bouygues E&S Contracting UK Ltd win prestigious RoSPA Gold Award
- Bouygues Energies & Services celebrate the grand opening of our new Scottish office.
- Bouygues Energies & Services engineers go back to school to inspire the next generation of engineers.
- Bouygues Energies & Services partner with Flybits to deliver the connected workplace of the future.
- Bouygues Energies & Services celebrate female engineers on international Women In Engineering Day.
- Bouygues Energies & Services win Considerate Constructors Scheme Bronze medal.
- Bouygues Energies & Services are showcasing at Data Centre World 2017
- We have delivered a £1.3M refurbishment at Barnet Hospital.
- Bouygues Energies & Services Partnering Approach with Biotechnology Stakeholders.
- We're proud to be on the CCS’ TMT2 Framework Agreement.
- Strengthening Supply Chain Partnerships Under the Wings of Concorde
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 104 105 106 107 108 109 110 111 112 113 114 115 116 117 118 119 120 121 122 123 124 125 126 127 128 129 130 131 132 133 134 135 136 137 138 139 140 141 142 143 144 145 146 147 148 149 150 151 152 153 154 155 156 157 158 159 160 161 162 163 164 165 166 167 168 169 170 171 172 173 174 175 176 177 178 179 180 181 182 183 184 185 186 187 188 189 190 191 192 193 194 195 196 197 198 199 200 201 202 203 204 205 206 207 208 209 210 211 212 213 214 215 216 217 218 219 220 221 222 223 224 225 226 227 228 229 230 231 232 233 234 235 236 237 238 239 240 241 242 243 244 245 246 247 248 249 250 251 252 253 254 255 256 257 258 259 260 261 262 263 264 265 266 267 268